- Home
- Large volume parental LVP ( Glass Bottle 100-500 ml )
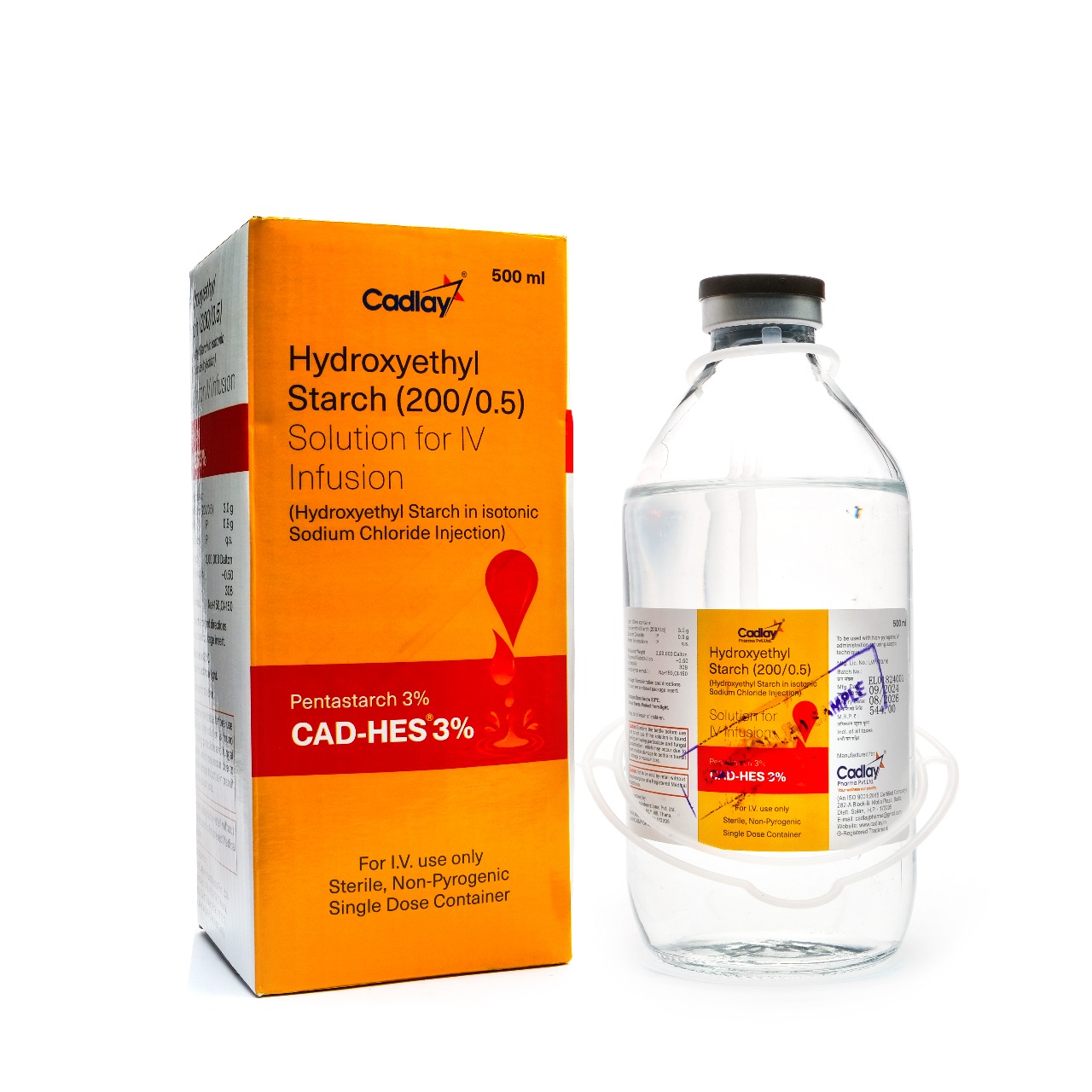
- Hydroxyethyl Starch Solution for IV Infusion
Hydroxyethyl Starch Solution for IV Infusion
(Hydroxyethyl Starch In Isotonic Sodium Chloride Injection)
Each 100ml Contains: Hydroxyethyl Starch (200/0.5) 3.0g Sodium Chloride IP 0.9g Water for Injections IP
Hydroxyethyl Starch Solution for IV Infusion – Hydroxyethyl Starch in Isotonic Sodium Chloride Infusion is a colloid solution used for intravenous fluid replacement in certain medical settings. Comprising hydroxyethyl starch particles suspended in isotonic sodium chloride, it expands blood volume, aiding in treating hypovolemia due to surgery, trauma, or other medical conditions. This solution temporarily increases blood pressure and maintains organ perfusion. However, its use has declined due to concerns about adverse effects, including renal impairment and coagulation abnormalities. Administration requires careful consideration of patient status, and healthcare professionals should weigh potential risks and benefits based on individual medical needs.
Benefits of Using Hydroxyethyl Starch IV Infusion
Hydroxyethyl Starch in Isotonic Sodium Chloride IV Infusion benefits intravascular volume expansion, aiding in treating hypovolemia resulting from surgery, trauma, or certain medical conditions. As a colloid solution, it helps restore and maintain blood volume, preventing complications associated with insufficient circulation. This can be crucial in situations requiring rapid fluid replacement, such as during surgeries or critical care. While historically utilized for its volume-expanding properties, it’s important to note that the use of HES has declined due to concerns about potential adverse effects. Healthcare professionals carefully assess individual patient needs and consider alternative options for fluid resuscitation.
How to Use Hydroxyethyl Starch IV Infusion?
Hydroxyethyl Starch in Isotonic Sodium Chloride Infusion is administered intravenously by healthcare professionals. The dosage and infusion rate depend on the patient’s clinical condition and fluid requirements. It is essential to follow the prescribed guidelines meticulously. During administration, vital signs and fluid balance should be closely monitored. Due to concerns about potential adverse effects, including renal dysfunction and coagulation abnormalities, healthcare providers must judiciously evaluate the need for and risks associated with Hydroxyethyl Starch. Its use is typically reserved for specific medical situations where intravascular volume expansion is critical, and alternative options have been carefully considered.
How Hydroxyethyl Starch IV Infusion Works?
Hydroxyethyl Starch in Isotonic Sodium Chloride IV Infusion works by expanding intravascular volume. Comprising starch molecules in a saline solution, it increases blood volume, improving cardiac output and tissue perfusion. HES molecules have a colloid osmotic effect, drawing water into the bloodstream, preventing fluid leakage from vessels. This helps treat hypovolemia resulting from surgery, trauma, or medical conditions. While historically used for its volume-expanding properties, its usage has declined due to concerns about adverse effects, including impaired renal function and coagulation abnormalities. Healthcare professionals must weigh the potential benefits against risks and consider alternative therapies based on patient needs.
Precautions While Using
When using Hydroxyethyl Starch in Isotonic Sodium Chloride IV Infusion, precautions are crucial. Due to concerns about renal impairment and coagulation abnormalities, healthcare providers must assess patient suitability carefully. Avoid its use in patients with renal dysfunction, sepsis, or coagulation disorders. During administration, continuous monitoring of vital signs, fluid balance, and renal function is essential. Consider alternative fluid therapies, especially in critically ill or septic patients. Adhere strictly to the recommended dosage and infusion rate guidelines. Promptly address any signs of adverse effects, such as changes in urine output or coagulation parameters. The decision to use HES should be individualized, emphasizing close medical supervision.
Side Effects
Hydroxyethyl Starch in Isotonic Sodium Chloride IV Infusion might lead to various side effects. Renal impairment is a significant concern, necessitating vigilant monitoring of kidney function. Coagulation abnormalities, such as bleeding or clotting issues, can occur. Anaphylactic reactions, including allergic responses, might manifest. The use of HES has been associated with increased mortality and adverse renal outcomes in certain patient populations. Due to these risks, its use has declined, and alternative fluid resuscitation strategies are often favored. Healthcare professionals must carefully weigh the potential benefits against these serious side effects, emphasizing individualized patient assessment and close monitoring.
| COMPOSITION | Each 100ml Contains: Hydroxyethyl Starch (200/0.5) 3.0g Sodium Chloride IP 0.9g Water for Injections IP |
|---|---|
| PACK SIZE | 100 ml |